Forschung AG Geiss-Friedlander (engl.)
|
Research: Molecular mechanisms of proteases in signaling and stress-response |
|
Research in the lab focuses on the molecular mechanisms of proteases in cell signaling and in response to stress. The human genome encodes more than 500 different proteases, that are involved in multiple biological pathways. Deregulation of protease activity or expression is linked to pathophysiological processes including neurodegenerative diseases, inflammation and cancer. Consequently, proteases have gained much attention as potential drug targets. |
|
While some proteases are dedicated to protein degradation, others act as sharp scissors cleaving their substrates at unique sites (limited proteolysis) thereby increasing the number of different protein-forms (proteoforms). |
|
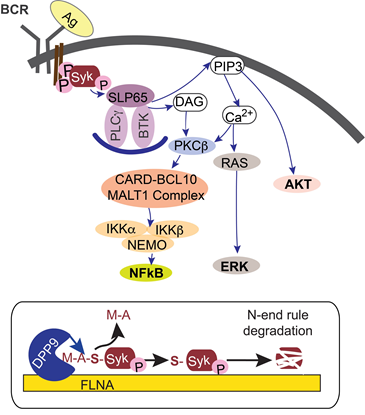
Two intracellular proteases Dipeptidyl peptidase 8 (DPP8) and DPP9 are two examples of proteases acting via limited proteolysis in which they cleave off dipeptides from the N-terminus of their substrates (preferentially post-proline) to produce new proteoforms. Through their unique ability to cleave a peptide bond following a proline residue, they present important points for regulation. DPP8 and DPP9 have been linked to the immune system and to cancer development, however the underlying molecular mechanisms leading to these outcomes are poorly understood. Research in the lab focuses on identifying the substrates and regulators of these proteases. By combining molecular biology, biochemistry in vitro and cell-based assays we study the outcome of substrate cleavage by DPPs and the regulation of these processes. Using such approaches, we identified the first endogenous substrate of DPP9, and showed that it plays a role in the maturation of antigenic peptides for presentation on MHC class I. Additionally, we demonstrated that DPP9 processes Syk, a key kinase in B cell receptor mediated signaling. By modifying its N-terminus, DPP9 exposes an N-degron in Syk, which targets the kinase for degradation by the N-degron pathway. Research in the lab focuses on the specific and overlapping roles of DPP8 and DPP9 in B cell signaling, cell survival and the cellular response to various stress conditions. Additional aspect involves the development of DPP8 and DPP9 inhibitors (‘Specific DPP8 and/or DPP9 inhibitors.’ WO2014068023, US9593148B2. Issued on March 14, 2017). |
 |
|
|

